Abstract
Salmonella spp. is one of the major foodborne pathogens responsible for causing economic losses to the poultry industry and bringing consequences for public health as well. Both the pathogen survival ability in the intestinal environment during inflammation as well as their relationship with the host immune system, play a key role during infections in poultry. The objective of this study was to quantify the presence of the macrophages and CD4+/CD8+ cells populations using the immunohistochemistry technique, in commercial lineages of chickens experimentally infected by wild-type and mutant strains of Salmonella Enteritidis and Salmonella Typhimurium lacking ttrA and pduA genes. Salmonella Enteritidis ∆ttrA∆pduA triggered a higher percentage of the stained area than the wild-type, with exception of light laying hens. Salmonella Typhimurium wild-type strain and Salmonella Typhimurium ∆ttrA∆pduA infections lead to a similar pattern in which, at 1 and 14 dpi, the caecal tonsils and ileum of birds showed a more expressive stained area compared to 3 and 7 dpi. In all lineages studied, prominent infiltration of macrophages in comparison with CD4+ and CD8+ cells was observed. Overall, animals infected by the mutant strain displayed a positively stained area higher than the wild-type. Deletions in both ttrA and pduA genes resulted in a more intense infiltration of macrophages and CD4+ and CD8+ cells in the host birds, suggesting no pathogen attenuation, even in different strains of Salmonella.
Introduction
Salmonella enterica is a foodborne pathogen that provokes losses to livestock as well as impacts directly on public health. Salmonella enterica subsp. enterica serovar Enteritidis (Salmonella Enteritidis) and Typhimurium (Salmonella Typhimurium) have been mainly associated with food infections for decades. Between 1995 and 2010, Salmonella Enteritidis was identified in 34,2% of all samples positive for Salmonella spp1. Besides that, Winter et al.2 published a substantial study investigating the tetrathionate-encoding gene role, choosing the serotype Salmonella Typhimurium but using mice as an experimental model. Taking this into account, deepening our knowledge of host–pathogen interaction could help to improve control and eradication measurements.
Several factors are involved in salmonellosis pathogenesis, such as the ability of the pathogen to replicate in an inflamed mucous environment, depending on nutrient acquisition and anaerobic respiration2. However, nutrient availability does not guarantee bacterial survival in a competitive environment densely populated by other microorganisms. Thus, the ability of Salmonella enterica metabolizes tetrathionate employing tetrathionate reductase to produce 1,2-propanediol as an energy source, confers a fitness advantage. This enzyme is constituted by TtrA, TtrB, and TtrC. The first subunit cited belongs to the molybdopterin (MPT) superfamily and has a FeS bounding domain that is involved in the reduction of tetrathionate into thiosulfate (S2O32−)2,3,4.
The 1,2-propanediol is used by bacterial microcompartments (MCP). This structure is constituted of seven different proteins, among which PduA is the major component of the MCP structure5. Firstly, 1,2-propanediol is converted to propionaldehyde, which in turn is reduced to propanol and propionate by propanediol dehydratase activity. This process generates ATP by phosphorylation, an electron (1-propanol) gradient to NAD regeneration, and an intermediary (propionyl-CoA) that can be used as carbon and energy source throughout methyl citrate via, being dependent on B12 vitamin synthesized in an endogenous manner6.
During chicken infections, the ability of Salmonella enterica serovars to invade and survive within the intestinal epithelial cells and macrophages is followed by immune response evasion7. In that context, the infection is a critical phase that depends on the interaction between bacterial and host cells and the bacterial ability to overcome the intestinal epithelium barriers to guarantee its colonization and replication. Nevertheless, it activates the inflammatory and immune responses8 that lead to endocytosis and phagocytosis by epithelial and antigen-presenting cells (APCs), respectively. The antimicrobial activity of these cells triggers an innate response via macrophages and the adaptive immune response assembly relies on CD4+ and CD8+ activation9.
To shed light on the host–pathogen interactions behind the intestinal infection by Salmonella in chickens, we evaluate the population of immune system cells during gut colonization and systemic infection in birds of commercial lineages challenged by wild-type mutant strains of Salmonella Enteritidis and Salmonella Typhimurium, carrying deletions in genes related to the metabolization of tetrathionate (ttrA) and 1,2-propanediol (pduA).
Results
Experiment 1—Salmonella Enteritidis challenge
The results of quantifying the presence of the immune response cells in caecal tonsils, caecum, ileum, and liver from broiler, light laying hens, and semi-heavy laying hens are shown in Tables 1, 2, and 3. We found more CD4+ and macrophages cells infiltration from broilers challenged with Salmonella Enteritidis ∆ttrA∆pduA (SEΔttrAΔpduA) than from broilers challenged with Salmonella Enteritidis wild-type strain (wt-SE) or non-infected birds, in all evaluated tissues. An exception was found for CD4+ cells infiltration at 3 dpi in the caecal tonsils, ileum, and liver, and for macrophages infiltration at 3 and 14 dpi in the liver from SEΔttrAΔpduA challenged broilers. Moreover, the number of CD8+ cells infiltrated was observed in a greater quantity from birds challenged with SEΔttrAΔpduA at 1 and 7 dpi in the caecum and liver, at 3 dpi in the caecal tonsils, and 14 dpi in the ileum. In contrast, wt-SE-challenged birds had high infiltration of CD8+ cells in the ileum and caecal tonsils at 1 and 7 dpi, respectively (Tab. 1; Supplementary Fig. S1).
Table 2, and Supplementary Fig. S2, show the CD4+ and CD8+, and macrophage infiltration found in tissues of semi-heavy laying hens. In general, there was great variation between the areas of cell infiltrates concerning both the challenge strain and the tissues studied. Birds challenged with SEΔttrAΔpduA showed higher areas covered by CD4+ cells (in ileum at 1 and 14 dpi, and liver at 7 dpi), and macrophages (in ileum at 7 dpi, and liver at 3, 7, and 14 dpi), than in the same tissue of wt-SE-challenged birds. On the other hand, CD8+ cells were found in larger amounts in caecal tonsils (at 1 and 7 dpi) and caecum (at 3 dpi) of birds challenged with wt-SE strain.
Differently observed in broiler and semi-heavy laying chicks, Salmonella Enteritidis ∆ttrA∆pduA triggered a less intense immune response cell areas than the wild type, in the challenge from light laying hens. Wt-SE-challenged birds showed larger CD4+ infiltration areas in caecal tonsils (at 1 and 3 dpi), caecum (at 3, 7, and 14 dpi), ileum (at 1 and 7 dpi), and liver (at 14 dpi). Similarly, challenges with SEΔttrAΔpduA have resulted in reduced infiltration areas of both CD8+ in caecal tonsils (at 1 dpi) and ileum (at 7 and 14 dpi), and macrophages in caecal tonsils and caecum (at 3 dpi), in comparison with wt-SE-challenged birds. No significant alterations of immune system cells area of CD8+ and macrophage cells were observed in the liver from both SEΔttrAΔpduA- and wt-SE-challenged birds. The detailed results of the challenge with light laying hens are shown in Table 3 (see Supplementary Fig. S3).
Experiment 2—Salmonella Typhimurium challenge
The results of quantifying the presence of the immune response cells in cecal tonsils, caecum, ileum, and liver from broilers, semi-heavy laying hens, and light laying hens are shown in Tables 4, 5, and 6, respectively. Overall, broilers infected by the mutant strain displayed a positive marked area higher than those challenged with Salmonella Typhimurium wild-type strain (wt-STM) strain, on all four sampling days. Moreover, no alterations of immune response cells were observed in the uninfected control groups.
The CD4+ cell areas in all tissues of broilers did reach a statistical difference at 1 dpi, with larger infiltrations for the challenge with Salmonella Typhimurium ∆ttrA∆pduA (STM∆ttrA∆pduA). A significant difference between the area of the quantified immune response from STM∆ttrA∆pduA- and wt-STM-challenged birds had been, with larger infiltrations when the challenge was with the mutant strain in the caecum and ileum at 1 dpi, and liver at 14 dpi (CD8+ cells); in the caecum, ileum, and liver at 1 and 14 dpi (macrophages) (Table 4; Supplementary Fig. S4).
The results from semi-heavy laying birds showed no statistical difference between mutant and wild-type strain challenges for CD4+ cells in the caecum, and CD8+ cells in the caecal tonsils and ileum in all 4 days post infections evaluated (Table 5; Supplementary Fig. S5). However, when a significative concentration area of immune system cells was observed the semi-heavy laying hens were challenged by STM∆ttrA∆pduA: major macrophages infiltration area in all tissues studied (at 1 and 14 dpi); a major infiltration area of CD4+ cells in the caecal tonsils and liver (at 1, 7, and 14 dpi) (Table 5; Supplementary Fig. S5).
Table 6, and Supplementary Fig. S6 show the CD4+, CD8+, and macrophage infiltration found in the tissues of light laying hens. Birds challenged with STM∆ttrA∆pduA showed major immune system cells area of CD4+ and macrophage cells in caecal tonsils and caecum (at 14 dpi), and ileum (at 1 and 14 dpi) in comparison to immune response cells area of the same tissues from wt-STM-challenged birds. No statistical difference for the CD8+ infiltration area had been found in the caecal tonsils, caecum, and ileum from challenged birds. In contrast to results obtained in other tissues, the liver from STM∆ttrA∆pduA-challenged birds had a more expressive stained area of immune response cells for CD4+ and CD8+ cells at all four dpi, and macrophages at 1 and 14 dpi.
Discussion
Bacteria, when exposed to anaerobic conditions, may use tetrathionate and 1,2-propanediol metabolic substrates for energy and respiration sources10. Thus, Salmonella spp. has long been subject to investigations into how the deletion of genes known to be responsible for these pathways would affect their survival in the host. To the best of our knowledge, only one study investigating simultaneously tetrathionate- and propanediol-encodings genes roles was published. Our research group reported the effects of these deletions by evaluating systemic infection and faecal excretion of Salmonella Enteritidis and Salmonella Typhimurium in commercial lineages of chicks11. To increase the discussion on this subject, the present results highlighted the immune cell infiltrated in different tissues of chick lineages challenged with both wild-type and mutant strains carrying deletions in ttrA and pduA genes.
Over the 2-week experiment the positively stained areas of CD4+ and CD8+ cells, and macrophages follow mostly a similar pattern, wherein at 1 and 14 dpi present a higher number of immune response cells. This can be explained by the primary contact of the host defense system when the pathogen invades. The previous report has shown that in chickens infected, even when Salmonella is not excreted at 12 dpi, the infection can become positive by cloacal swab from 13 dpi12, explaining why immune system cells areas at 3 and 7 dpi were lower but back to an increase.
At first glance, it would be expected that the elicited response of the host would be reduced when both pduA and ttrA were deleted since these genes play an important role in the survival during infection by Salmonella2,4,13,14,15. However, our results comparing a double mutant lacking both genes showed the opposite, the mutant strains of Salmonella Enteritidis and Salmonella Typhimurium triggered higher immune response cells than the wild types of strains. A shortest stained area could lead to a high number of colonies on the intestinal tract, corroborating a previous study, wherein Salmonella Enteritidis ∆ttrA∆pduA and Salmonella Typhimurium ∆ttrA∆pduA strains were recovered in higher numbers from cloacal swabs than their wild type correlated11.
Salmonella can behave as an extracellular or an intracellular bacterium, depending on the nutrient repertoire available, and occurs as a switch between intestinal colonization and internalization into host cells16. When the bacteria are ingested and killed by macrophages, some peptide fragments are transferred to the surface of the antigen-presenting cell, being encoded by the major histocompatibility complex (MHC), class II. This peptide–MHC II binding stimulates the T CD4+ lymphocytes. However, if the bacteria decide to invade the host cell, entering the cytoplasm of the macrophage, the peptide connection with another type of MHC, class I, stimulates T CD8+ lymphocyte production17.
Interestingly, CD4+ and CD8+ cells present the same pattern of the macrophages throughout the experiment, even representing different immune responses. Since CD4+ and CD8+ cells are mainly representing T lymphocytes which are part of the adaptive immune response, the macrophages, are part of the innate immune response9. In addition to this, we observed that broilers present more expressive positive marked area than laying hens, confirmed by a previous study where broilers challenged with mutant strains showed, for example, a more invasive intestinal colonization and systemic infection11.
Our findings suggest that the immunohistochemistry approach provides interesting information about the behavior of immune response cells on multiple organs of different commercial lineages during infection by Salmonella enterica serovars. Moreover, the present study evidence that deleting both genes, even in different strains of Salmonella, resulted in bacteria that elicited a higher immune response cell in the host, showing that the pathogen has not been attenuated. We can consider that perhaps Salmonella was able to find another survival mechanism becoming even more pathogenic. The utilization of ttr and pdu operons in consonance with cob and prp operons has been shown, in the previous study, necessary for anaerobic respiration16, leading us to believe that is not only required to delete more genes from each operon18, but we also have to ponder deleting this whole set, to reach less pathogenic strains of Salmonella enterica.
Materials and methods
The experiments, performed following relevant guidelines and regulations, were approved by the Ethical Committee on the Use of Animals of Sao Paulo State University (CEUA/Unesp Process—006621/18; on May 10th, 2018), were carried out in the Avian Pathology Laboratory of the Department of Pathology, Theriogenology, and One Health from the School of Agricultural and Veterinary Sciences, Sao Paulo State University (FCAV/Unesp), Jaboticabal, Brazil.
Bacterial strains and mutant construction
The bacterial strains used here were stored within a cryoprotectant medium compounded by Lysogeny broth (LB; BD DifcoTM, USA) supplemented with 30% of glycerol (Merck, BR—H30402394 228) and storage in an ultra-freezer (− 80 °C) at the Avian Pathology Laboratory from FCAV/UNESP. Salmonella Enteritidis P125109 (accession number: AM933172) and Salmonella Typhimurium str. 9819 were induced to nalidixic acid- and spectinomycin-resistance (NalrSpcr) and they provided the genetic background for constructing mutant strains by Lambda-red technique20 with minor modifications, described in Saraiva et al.11. Mutant bacterias constructed here are identified on the text as SEΔttrAΔpduA (Salmonella Enteritidis ∆ttrA∆pduA) and STMΔttrAΔpduA (Salmonella Typhimurium ∆ttrA∆pduA).
In vivo experiment
Experiment 1—Salmonella Enteritidis
Thirty-six 1-day-old chicks from each of three different lineages (broiler, semi-heavy laying hens, and light laying hens), totaling one hundred and eight animals, were obtained from commercial hatcheries. At arrival, the bottom of transport card boxes was examined to confirm the Salmonella-free status of the birds21, and the animals were housed within metallic cages inside the acclimatized room and received antibiotic-free feed and water ad libitum. A 24-h light program was chosen on the first day to ensure optimal water and food ingestion, then a 12-h light program was adopted in the first week, decreasing to 8 h on the remaining days.
The inoculum was prepared according to Berchieri Junior et al.22. For this, the frozen cultures were inoculated in LB and incubated overnight at 37 °C under 150 rpm. On the following day, the bacterial cultures were transferred into fresh media and incubated for 18 h under the same conditions as previously. Then, 0.2 mL from the cultures containing 108 colony-forming units per mL (CFU/mL) were orally inoculated by metallic gavage directly into the birds’ crop.
Nine groups were formed (A to I) and randomly divided according to the different lineages and strains (Table 7). At one-, three-, seven-, and 14-days post-infection (dpi), three birds per group each day, by morning, were euthanized by cervical dislocation to harvest the medial section of caecal tonsils, caecum, and ileum, and the distal section of the liver left lobe for further immunohistochemistry (IHC) analysis. For this, samples were submerged within n-Hexane p.a. (n-Hexano p.a., Synth, Brazil) previously refrigerated in liquid nitrogen. Immediately after the tissue freezing, it was transferred into a 2 mL cryotube (Corning, USA) and conditioned in liquid nitrogen. After sampling, the tissues were stored at − 80 °C until the process for IHC.
Experiment 2—Salmonella Typhimurium
This experiment was carried out following the same characteristics mentioned above in experiment 1. Thirty-six chicks (1 day old) were randomly divided into nine groups (A to I) based on their lineages and strains (Table 7).
Immunohistochemistry
Tissue section
The collected samples were transferred from − 80 °C to cryostat (Leica CM1860, Leica Biosystems Nussloch GmbH, Germany) at − 22 °C where were individually blocked in O.C.T. compound (Tissue-Tek®, Sakura Finetek Europe B.V., Netherlands) per 30 min prior to 6 µm section using low profile disposable blades (Leica 819, Leica Biosystems Nussloch GmbH, Germany). It is noteworthy that sections were done at − 22 °C, except liver sections, which were done at − 15 °C. Slides containing three repetitions of the sectioning of each organ per immune cell response marked were prepared, with thinning between each repetition. Using a paintbrush, the tissue section cuts were placed on histological slides pre-treated with poly-l-lysine (Sigma-Aldrich, United Kingdom, Cat no. P4832) e silane (Sigma-Aldrich, USA. Cat no. 440574). The slides were stored at − 20 °C thereafter until IHC staining.
Immune cells staining
Firstly, the slides were submerged in 200 mL refrigerated acetone (Acetone P.A.—A.C.S., Synth, Brazil) and incubated at − 20 °C for 10 min. After that, the slides were transferred into a humidity chamber (EasyPath®, Brazil) at room temperature for 5 min to dry the samples. The slides were washed thereafter with PBS and a puddle was left for 5 min to avoid tissue dehydration. Then, tissues were submerged in 200 mL of 4% H2O2 per 10 min in a dark place and washed again with PBS. The area around the tissue sections was dried with absorbent paper and the sample was bypassed by a hydrophobic pen (Dako Pen, Dako Denmark A/S, Denmark). The washing step leaving a puddle was repeated as previously.
The biotin-free kit Mouse and Rabbit Specific HRP/DAB IHC Detection—Micropolymer (Abcam©, USA) was used to stain the immune cells, choosing the Avidin–Biotin Streptavidin Peroxidase Complex (ABC) method. For this, the puddle was removed, and non-specific background color blocker reagent droplets were added to the tissues. The slides were maintained inside the humidity chamber in a dark place for 30 min, the washing step was repeated, and 200 µL of primary antibody (Mouse Anti-Chicken CD4-UNLB; Mouse Anti-Chicken CD8α-UNLB; Mouse Anti-Chicken Monocyte/Macrophage-UNLB, Southern Biotech, USA) diluted in a proportion of 1:200 (v/v) in the antibody diluent reagent (Antibody diluent, Abcam©, USA) was added thereafter. The slides were incubated at 4 °C for 18 h.
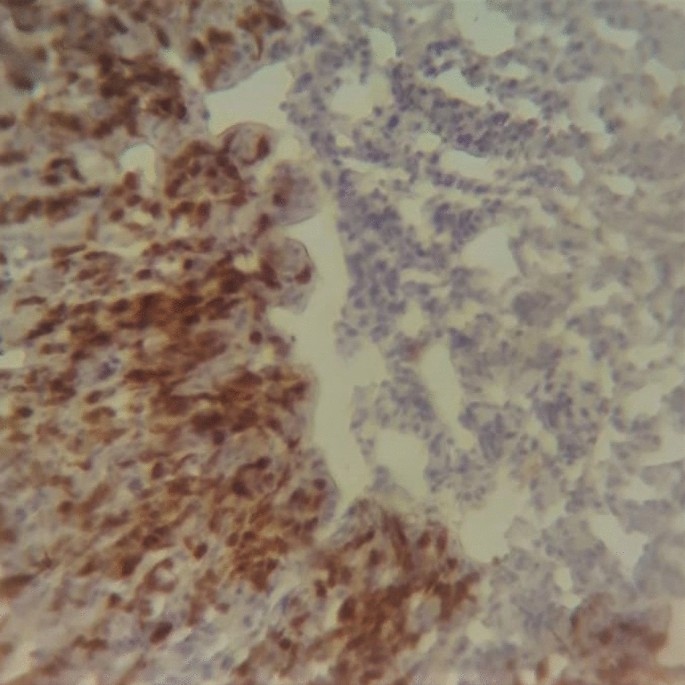
On the following day, the slides were washed as previously. Then, droplets of the secondary antibody (Reveal Complement, Abcam©, USA) was added after removing the excess PBS and the humidity chamber was placed in a dark ambient for 30 min. Subsequently, a drop of 3,3′-diaminobenzidine (DAB Chromogen 50 ×, Abcam©, USA) was diluted in 1 mL of substrate (DAB Substrate, Abcam©, USA), which volume is enough for three slides, and added to the tissue sections. One minute later, the slides were submerged in 200 mL dH2O for 5 min. After that, they were transferred to a plastic cube containing Harris hematoxylin (Êxodo Científica, Brazil) and were left for 1 min. Posteriorly, the slides were washed for 10 min under running water at low pressure. The slides were submitted to the alcohol-xylene series (70% Alcohol, 90% Alcohol, 100% Alcohol, Xylene I, and Xylene II). In the end, the coverslips were placed on the slides after adding a drop of water-free mounting medium (Entellan®, Merck, Brazil). Images from the tissue sections were taken randomly, choosing five random view fields, using an optical microscope (lens 400 ×) (Coleman®, model N-120) with a digital camera adapter, for further statistical analysis (Fig. 1).
Data analysis
The percentage of CD4+ and CD8+ cells and macrophages was calculated using Image-Pro Plus v.4.5.0.29 (MediaCybernetics, USA). They were quantified as percentage values by the immune cell marker positive area/total area. Statistical analysis and graphics were done using the software GraphPad Prism v.8.0.1 for macOS (GraphPad Software, La Jolla California, USA) and data were submitted to Variance Analysis (ANOVA) followed by Bonferroni multiple comparisons, considering a significance level lower than 5% (P ≤ 0.05).
Ethical statement
The authors declare that all the in vivo experiment was performed in complete accordance with relevant guidelines and regulations. Moreover, the authors declare that the study was carried out in accordance with ARRIVE guidelines (https://arriveguidelines.org).
Data availability
The data supporting this study’s findings are available from the corresponding author, upon reasonable request.
References
-
Freitas Neto, O. C., Penha Filho, R. C. & Barrow, P. A. Sources of human non-typhoid salmonellosis: A review. Braz. J. Poult. Sci. 12(1), 1–11. https://doi.org/10.1590/S1516-635X2010000100001 (2010).
-
Winter, S. E. et al. Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature 467, 426–429. https://doi.org/10.1038/nature09415 (2010).
-
Hinsley, A. P. & Berks, B. C. Specificity of respiratory pathways involved in the reduction of sulfur compounds by Salmonella enterica. Microbiology (Reading) 148, 3631–3638. https://doi.org/10.1099/00221287-148-11-3631 (2002).
-
Thiennimitr, P. et al. Intestinal inflammation allows Salmonella to use ethanolamine to compete with the microbiota. Proc. Natl. Acad. Sci. U.S.A. 108, 17480–17485. https://doi.org/10.1073/pnas.1107857108 (2011).
-
Staib, L. & Fuchs, T. M. Regulation of fucose and 1,2-propanediol utilization by Salmonella enterica serovar Typhimurium. Front. Microbiol. 6, 1116. https://doi.org/10.3389/fmicb.2015.01116 (2015).
-
Horswill, A. R. & Escalante-Semerena, J. C. Propionate catabolism in Salmonella Typhimurium LT2: Two divergently transcribed units comprise the prp locus at 8.5 centisomes, prpR encodes a member of the sigma-54 family of activators, and the prpBCDE genes constitute an operon. J. Bacteriol. 179, 928–940. https://doi.org/10.1128/jb.179.3.928-940.1997 (1997).
-
Soria, M. C., Soria, M. A., Bueno, D. J. & Terzolo, H. R. Comparison of 3 culture methods and PCR assays for Salmonella Gallinarum and Salmonella Pullorum detection in poultry feed. Poult. Sci. 92, 1505–1515. https://doi.org/10.3382/ps.2012-02926 (2013).
-
Van Immerseel, F. et al. Dynamics of immune cell infiltration in the caecal lamina propria of chickens after neonatal infection with a Salmonella Enteritidis strain. Dev. Comp. Immunol. 26, 355–364. https://doi.org/10.1016/s0145-305x(01)00084-2 (2002).
-
Montassier, H. J. Fisiopatologia do sistema immune. In Doenças das Aves (eds AndreattiFilho, R. L. et al.) 467–489 (FACTA, 2020).
-
Price-Carter, M., Tingey, J., Bobik, T. A. & Roth, J. R. The alternative electron acceptor tetrathionate supports B12-dependent anaerobic growth of Salmonella enterica serovar Typhimurium on ethanolamine or 1,2-propanediol. J. Bacteriol. 183, 2463–2475. https://doi.org/10.1128/JB.183.8.2463-2475.2001 (2001).
-
Saraiva, M. et al. Deciphering the role of ttrA and pduA genes for Salmonella enterica serovars in a chicken infection model. Avian Pathol. https://doi.org/10.1080/03079457.2021.1909703 (2021).
-
Beal, R. K., Wigley, P., Powers, C., Barrow, P. A. & Smith, A. L. Cross-reactive cellular and humoral immune responses to Salmonella enterica serovars Typhimurium and Enteritidis are associated with protection to heterologous re-challenge. Vet. Immunol. Immunopathol. 114, 84–93. https://doi.org/10.1016/j.vetimm.2006.07.011 (2006).
-
Winter, S. E. & Bäumler, A. J. A breathtaking feat: To compete with the gut microbiota, Salmonella drives its host to provide a respiratory electron acceptor. Gut Microbes 2, 58–60. https://doi.org/10.4161/gmic.2.1.14911 (2011).
-
Rivera-Chávez, F. I. et al. Salmonella uses energy taxis to benefit from intestinal inflammation. PLoS Pathog. 9, e1003267. https://doi.org/10.1371/journal.ppat.1003267 (2013).
-
Khan, C. M. The Dynamic Interactions between Salmonella and the microbiota, within the challenging niche of the gastrointestinal tract. Int. Sch. Res. Not. 2014, 1–23. https://doi.org/10.1155/2014/846049 (2014).
-
Yoo, W., Kim, D., Yoon, H. & Ryu, S. Enzyme IIANtr regulates Salmonella invasion via 1,2-propanediol and propionate catabolism. Sci. Rep. 7, 44827. https://doi.org/10.1038/srep44827 (2017).
-
Salyers, A. A. & Whitt, D. D. Host defenses against bacterial pathogens: Defenses of tissue and blood. In Bacterial Pathogenesis: A Molecular Approach (eds Salyers, A. A. & Whitt, D. D.) 16–29 (ASM Press, 1994).
-
Góes, V. et al. Salmonella Heidelberg side-step gene loss of respiratory requirements in chicken infection model. Microb Pathog. 171, 105725. https://doi.org/10.1016/j.micpath.2022.105725 (2022).
-
Barrow, P. A., Hassan, J. O. & Berchieri, A. Jr. Reduction in faecal excretion of Salmonella Typhimurium strain F98 in chickens vaccinated with live and killed S. Typhimurium organisms. Epidemiol. Infect. 104, 413–426. https://doi.org/10.1017/s0950268800047439 (1990).
-
Datsenko, K. A. & Wanner, B. L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. U.S.A. 97, 6640–6645. https://doi.org/10.1073/pnas.1201632977 (2000).
-
Zancan, F. B., Berchieri Junior, A., Fernandes, S. A. & Gama, N. M. S. Q. Salmonella spp. investigation in transport box of day old birds. Braz. J. Microbiol. 31, 230–232. https://doi.org/10.1590/S1517-83822000000300016 (2000).
-
Berchieri, A. Jr., Murphy, C. K., Marston, K. & Barrow, P. A. Observations on the persistence and vertical transmission of Salmonella enterica serovars Pullorum and Gallinarum in chickens: Effect of bacterial and host genetic background. Avian Pathol. 30, 221–231. https://doi.org/10.1080/03079450120054631 (2001).
Acknowledgements
Sao Paulo Research Foundation (FAPESP) and Coordination for the Improvement of Higher Education Personnel (CAPES) research grants are gratefully acknowledged. The authors wish to thank the Coordination for the Improvement of Higher Education Personnel [grant number CAPES 88887.512107/2020-00 (J.M. Cabrera)], Sao Paulo Research Foundation [grant number FAPESP 2018/2130-2 (M.M.S. Saraiva), grant number FAPESP 2020/06076-2 (D.F.M. Monte), and grant number FAPESP 2018/03189-0 (A. Berchieri Junior)]. We are in debt to Prof. Luciane Helena Gargaglioni Batalhão for kindly providing the use of cryostat (Leica CM1860, Leica Biosystems Nussloch GmbH, Germany).